On March 13, 2026, the research team led by Academician Hefeng Huang, Academician of the Chinese Academy of Sciences, from the Center for Reproductive Medicine at Zhejiang University’s International Institute of Medicine (ZJU-IIM), published an importantsignificant study online in the journal Vita, titled “TRPV4 acetylation in prenatal liver prevents low glucose-induced inhibition of mTORC1 and safeguards fetal development”.
Glucose isserves as a criticalvital energy source for fetal growth and development. However, since fetal blood glucose levels are naturally lower than those of the mother. ,This raises a fundamental scientificbiological question arises: how does the fetal liver, an organ characterized by highly active in anabolic metabolism, mainsustain robust protein synthesis under this low-glucose environment, which typically inhibits that would normally anabolic processes?
The study revealed that fetal liver cells maintain high proliferation capacity even in glucose-free culture condition. This remarkable capability ismedia, a unique trait absent in adult liver cells orand commonly used standard cell lines (e.g., HEK293T), and this adaptation vanishedisappears within 24 hours after birth.
Further investigation uncovered the underlying mechanism. Mechanistically, while Under low glucose conditions, typically closesmost TRPV channels close to inhibitand supress the lysosomal glucose-sensing pathway. In contrast,, TRPV4 remains persistently open in fetal liver cells. Further mMechanistic investigations identified that acetylation at lysine 608 (K608) of TRPV4, catalyzed by the acetyltransferase p300, is the core molecular basis underlying this insensitivity to low glucose.
In mouse models,L liver-specific knockoutdisruption of TRPV4 K608 acetylation in mice resulted in diminishled to reduced mTORC1 activity, severe liver developmental abnormalities, and both prenatal- and postnatal lethality. Siminar, a phenotypes that could be recapitulated were produced by treating wild-type embryos with the mTORC1 inhibitor Rrapamycin.
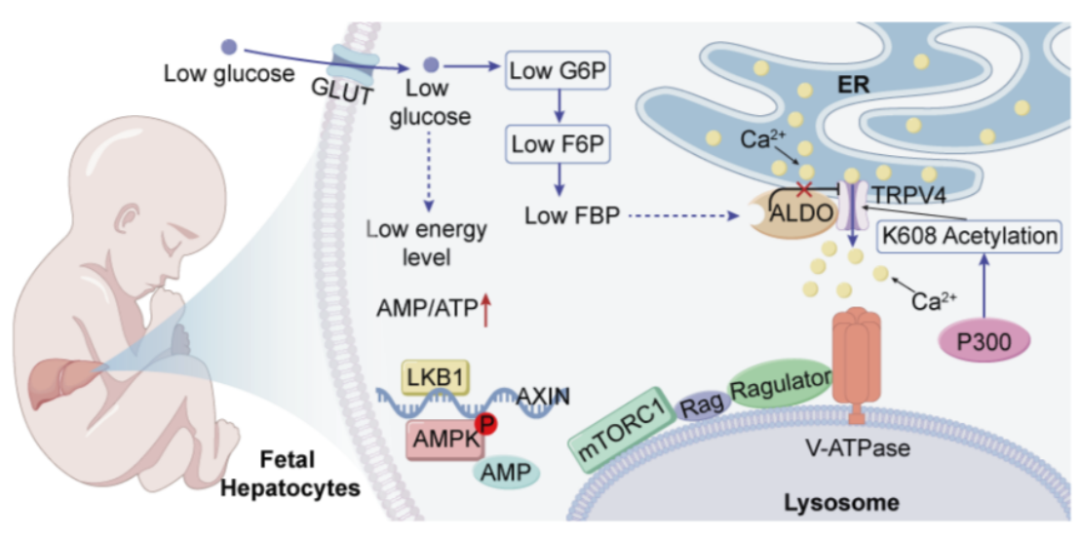
Overall, this study elucidates a noveluncovers a previously unknown, developmentally restricted, fetal-specific metabolic adaptation mechanism unique in the fetal liver. These findings, offering provide criticaimportantl insights into the regulatoryion mechanism of fetal development and potential implications for understanding the link between prenatal nutritional programming and the risk of developing metabolic diseases later in lifein adulthood.
Link to the full article:
https://www.vita-journal.com/vita/EN/10.15302/vita.2026.03.0017